Compositions and Methods for Treating and Preventing Obesity
This invention provides a method for modulating the energy consumption pathways in adipocytes, involving ARID5B, genetic variant rsl421085, IRX3, and IRX5. In addition, this invention allows for the identification of subjects at risk of developing obesity or disorders mediated by a dysregulation of the energy consumption pathway.
Researchers
-
compositions and methods for manipulation of adipocyte energy consumption regulatory pathway
United States of America | Granted | 10,774,326
Figures
Technologies
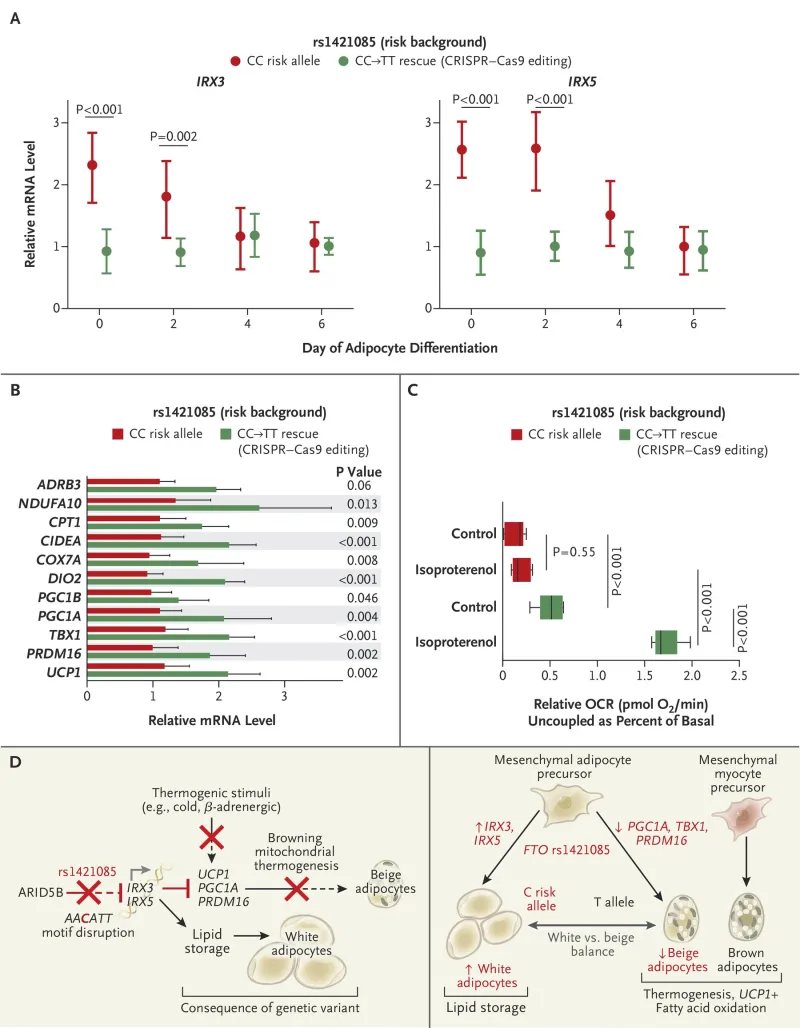
This invention provides a method for manipulating the energy consumption circuitry in adipocytes to reverse their obesity phenotype, or stimulate their obesity phenotype, in a cell-autonomous way. This technology involves contacting adipocytes with a sequence targeted therapeutic that modifies the expression of FTO obesity- associated locus and the causal variant that recapitulates the molecular and cellular signatures of obesity, the genetic variant rsl421085. Modification of the rs 1421085 of the FTO locus disrupts a conserved motif for the repressor ARID5B in risk allele carriers, activating of a potent mesenchymal adipocyte progenitor enhancer in the FTO locus.
This results in an increased expression of IRX3 and IRX5 in primary adipocyte progenitors, leading to a cell-autonomous developmental shift from energy-dissipating beige (brite) adipocytes to energy-storing white adipocytes, with a reduction in mitochondrial thermogenesis as well as an increase in lipid storage.
Problem Addressed
Obesity affects more than 500 million people worldwide and is one of the leading contributors to common and severe disorders, including type 2 diabetes, cardiovascular disorders, and cancer. Considerable efforts have been made to define its causal mechanisms, as understanding the genetic cause of obesity could illuminate the causal genetic mechanism of other disorders. The fat mass and obesity- associated (FTO) locus has been shown to possess the strongest association with obesity risk. Despite considerable efforts to define causal mechanisms, the functional variant remains uncharacterized among 82 highly-associated single nucleotide variants (SNVs), and the molecular and cellular intermediate phenotypes, mechanisms, and biological processes leading to obesity remain unknown.
The present invention provides a method of modulating energy consumption (e.g., thermogenesis) in an adipocyte, comprising contacting the adipocyte with a genome editing system that modifies the FTO locus. This invention has the potential to identify patient with obesity risk, as well to uncover therapeutic targets for adipocyte-autonomous pharmacological treatment for anti-cachexia and anti-obesity effects.
Advantages
- Direct targeting of the fat mass and obesity- associated (FTO) locus genetic variant, rs 1421085
- Diagnosis tool for subjects at risk of developing obesity or with a dysregulation of the energy consumption pathway
- Potential to uncover pharmacological targets for anti-cachexia and anti-obesity related diseases
Publications
Claussnitzer, M., et al. (2015). FTO Obesity Variant Circuitry and Adipocyte Browning in Humans. New England Journal of Medicine, 373, 895-907. doi: 10.1056/NEJMoa1502214.
License this technology
Interested in this technology? Connect with our experienced licensing team to initiate the process.
Sign up for technology updates
Sign up now to receive the latest updates on cutting-edge technologies and innovations.