Adventitious Agent Clearance by Use of Spiral Inertial Microfluidics
This technology is related to cases #26458, #21685, #20884, #18940, #15955, #15591, #14462 as well as #18989, #16927
This invention discloses a spiral inertial microfluidics–based sorting device that selectively removes adventitious agents (AAs)–including bacteria, viruses, and mycoplasma–from cells that produce therapeutic products such as enzymes and antibodies, as well as from cells that constitute therapeutic products themselves. This technology leverages size-dependent differences in inertial lift and secondary Dean flow forces to position particles within a spiral microfluidic channel and continuously separate smaller AAs from larger cells. By simplifying flow control and addressing the size-range limitations of previous spiral microfluidic designs, this invention provides a single-inlet solution for the selective removal of AAs from therapeutic cell materials.
Researchers
-
spiral inertial microfluidic devices and methods to remove contaminants
United States of America | Published application
Figures
Technology
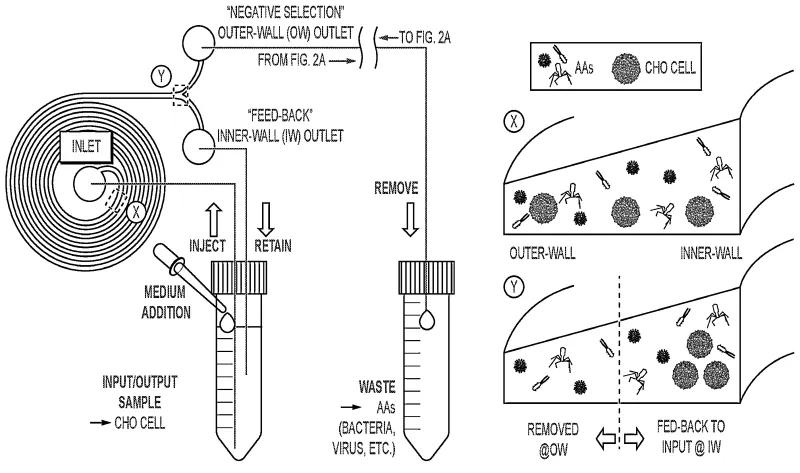
The spiral microfluidics sorter operates continuously in a closed feedback loop to remove AAs while retaining cells. First, the input sample is loaded into a container and then injected into the spiral microfluidic channel. At the beginning of the spiral microchannel, all cells and particles are randomly dispersed. The microchannel features a taller inner wall and a shorter outer wall, creating an asymmetric geometry that modifies the local magnitude of inertial lift and Dean drag forces to separate particles as they flow through the device. As fluid travels through the spiral, larger cells experience strong inertial lift forces which drive them toward a stable equilibrium are transported to the inner-wall (IW) side of the spiral microchannel. Smaller AAs experience Dean drag forces which cause them to remain randomly dispersed within the channel. At the end of the spiral microchannel, a bifurcation separates the dispersed AAs through the outer-wall (OW) outlet, while the cell-rich IW outlet is fed back to the system input. The bifurcation ratio can be tuned to maximize cell retention, and fresh medium is continuously added to replace the volume lost through the OW waste stream. After a sufficient number of cycles, AAs are progressively removed while the majority of cells are retained within the IW feedback loop.
Problem Addressed
The production of biologic therapies can be compromised when AAs such as bacteria, viruses, and mycoplasma enter cellular materials, including cell banks used for therapeutic protein production and cell therapies. Existing techniques to separate particulate matter from fluids such as centrifugation, fluorescence‑activated cell sorting, and magnetic‑activated cell sorting are often labor‑intensive, harsh on cells, or require complex labeling steps. Conventional spiral microfluidic solutions have also been constrained by two‑inlet (sheath‑flow) designs which increase control complexity, and by geometries that struggle to reliably separate particles in the intermediate confinement‑ratio, reducing reliability and operating flexibility in real bioprocess settings. This invention addresses these challenges by providing a simple, single‑inlet spiral device that maintains high throughput, uses a spatially compact design, and operates without additional force fields to enable the selective removal of AAs from therapeutic cell banks.
Advantages
- Supports closed, automated operation to reduce manual handling and risk of contamination
- Allows continuous operation until contaminants are removed to a satisfactory level while retaining cells of interest
- Can replace labor-intensive centrifugation and washing steps
- Enables continuous, clog-free operation by avoiding membrane-based filtration
- Prevents cell aggregation, supporting recovery of viable, planktonic cells
- Can be re-used many times with proper device washing procedures
License this technology
Interested in this technology? Connect with our experienced licensing team to initiate the process.
Sign up for technology updates
Sign up now to receive the latest updates on cutting-edge technologies and innovations.