Novel Peptidic Agents for Targeting Chemoresistant Anti-Apoptotic Bfl-1
Peptide inhibitors of Bfl-1, an anti-apoptotic protein, allow for selective targeting of Bfl-1 dependent cancer cells in therapeutic treatment. Selective peptides may also be used as Bfl-1-detection reagents. This allows for cellular profiling for anti-apoptotic protein dependency, thus potentially acting as a diagnostic tool for stratifying patients.
Researchers
-
selective bfl-1 peptides
United States of America | Granted | 11,198,715
Figures
Technology
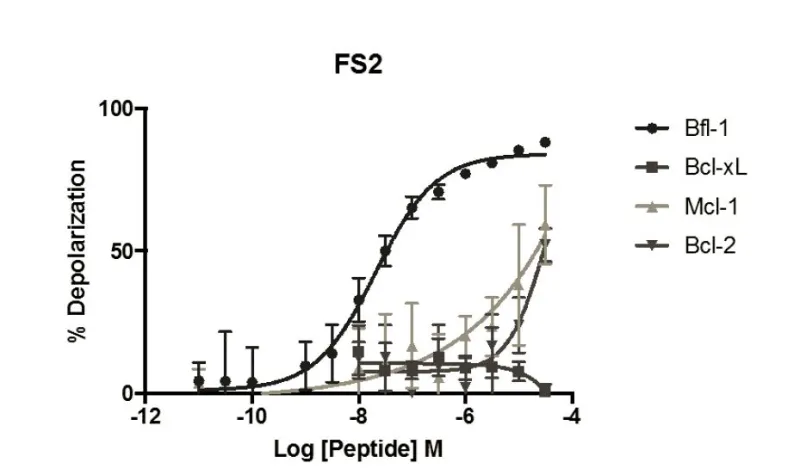
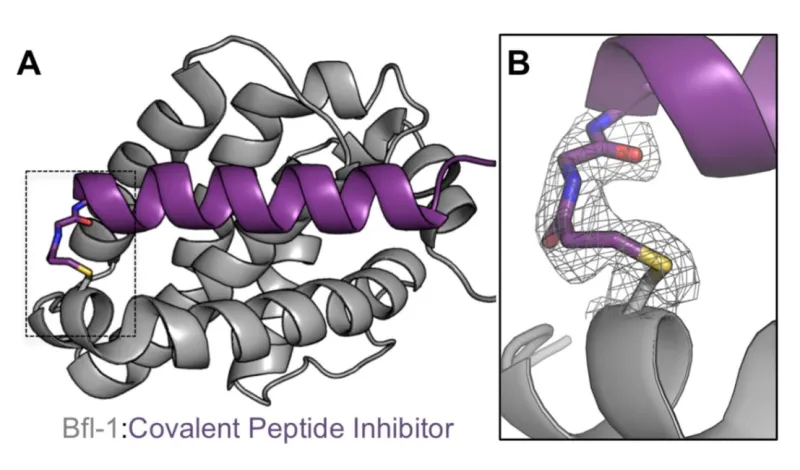
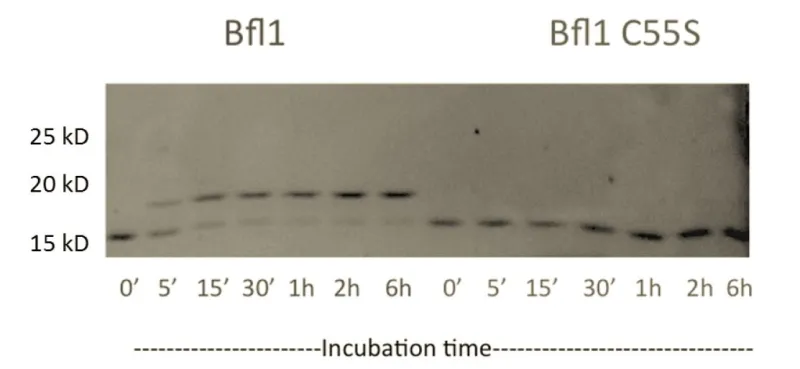
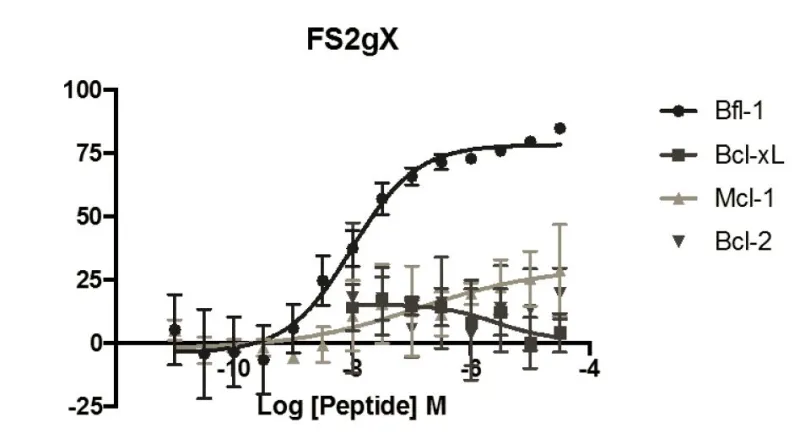
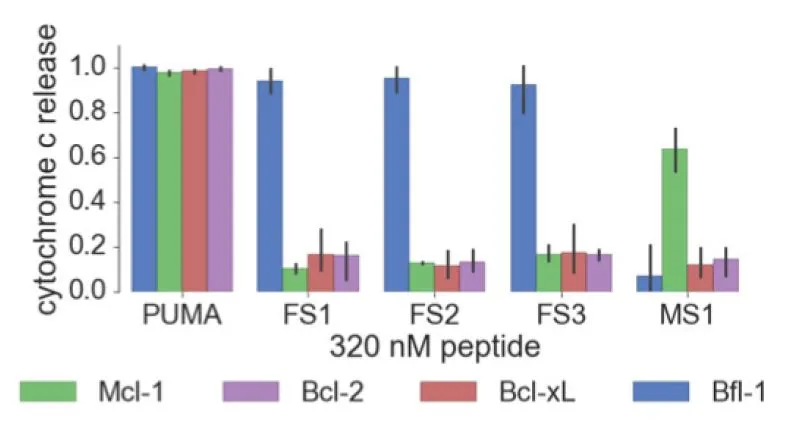
Selective peptide inhibitors of Bfl-1 were identified by screening for sequence variants of the BH3 motif of PUMA, a highly potent binder of Bcl-2 family members. Peptides were designed using computational analysis followed by experimental screening that identified peptides with high affinity for Bfl-1 yet weakened interactions with other anti-apoptotic proteins. The lead peptide inhibitors are >100-fold selective for binding to Bfl-1 binding over other anti-apoptotic proteins of Bcl-2 family. Selectivity towards Bfl-1 was further augmented by modifying the peptide inhibitors with electrophiles to allow for covalent interaction in the binding pocket of Bfl-1. The selectivity of the peptide Bfl-1 inhibitors in a cellular context was confirmed by the relative mitochondrial depolarization or cytochrome-c release after peptide treatment of Bfl-1 dependent cells vs. other anti-apoptotic protein dependent cells.
Problem Addressed
Many cancer cells overexpress anti-apoptotic Bcl-2 family proteins that confer resistance to chemotherapy treatment. Selective inhibitors of Bcl-2 are available in clinic to treat Bcl-2 dependent cancers. However, such tools are missing for Bfl-1. Selective inhibitors allow for selective targeting of Bfl-1 dependent cancer cells while limiting systemic toxicity.
Advantages
- The peptide inhibitors are potent and selective towards Bfl-1 with >100-fold selectivity over other anti-apoptotic proteins
- The peptide inhibitors can also be used as diagnostic tools to profile cells for Bfl-1 dependency
Publications
Jenson, J. M., et al. "Epistatic mutations in PUMA BH3 drive an alternate binding mode to potently and selectively inhibit anti-apoptotic Bfl-1." eLife 6 (2017): e25541. https://doi.org/10.7554/eLife.25541.
License this technology
Interested in this technology? Connect with our experienced licensing team to initiate the process.
Sign up for technology updates
Sign up now to receive the latest updates on cutting-edge technologies and innovations.